Report by
Outbreak
Listeria Outbreak investigation linked to Frozen Supplemental Shakes
1 year ago •source fda.gov
United States
####Update May 16th, 2025:The FDA and CDC, alongside state and local partners, have concluded their investigation into a multistate outbreak of Listeria monocytogenes infections linked to Lyons ReadyCare and Sysco Imperial frozen supplemental shakes. These products were manufactured by Prairie Farms Dairy, Inc. at their facility in Fort Wayne, Indiana.
In response to the outbreak, Lyons Magnus LLC voluntarily recalled 4 oz. Lyons ReadyCare and Sysco Imperial frozen supplemental shakes with best by dates from February 21, 2025 to February 21, 2026. As of this update, no recalled products should remain on the market.
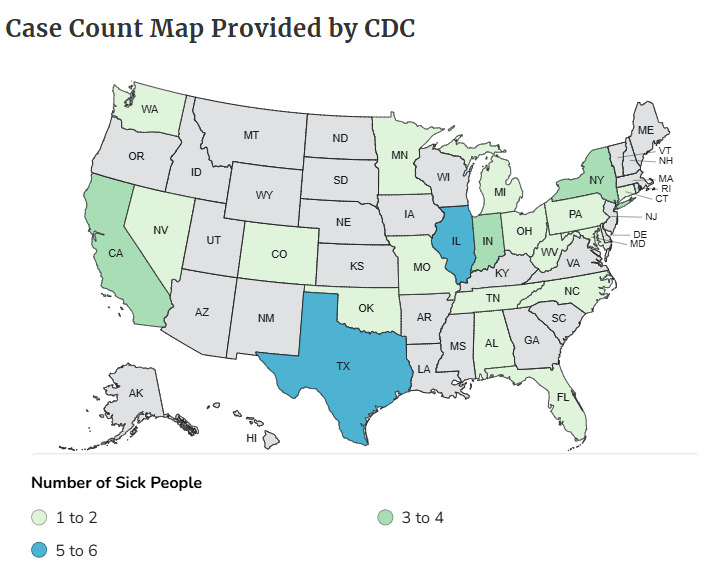
Final Outbreak Summary:
- Total illnesses: 42
- Hospitalizations: 41
- Deaths: 14
- States affected: 21 (including AL, CA, CO, CT, FL, IL, IN, MD, MI, MN, MO, NC, NV, NY, OH, OK, PA, TN, TX, WA, and WV)
- Last reported illness onset: March 13, 2025
- Product distribution: Nationwide
The outbreak is now considered over, and the FDA has officially closed its investigation.
Source: FDA
Update February 24th:
The FDA and CDC, in partnership with state and local officials, continue to investigate a multistate outbreak of Listeria monocytogenes infections linked to Lyons ReadyCare and Sysco Imperial Frozen Supplemental Shakes. Prairie Farms Dairy, Inc., manufactured these products at its facility in Fort Wayne, Indiana.
As of February 24, 2025, a total of 38 cases have been reported across 21 states, with 37 hospitalizations and 12 deaths. Many of the affected individuals were residents of long-term care facilities.
During the investigation, the FDA conducted an inspection at the Prairie Farms facility and collected environmental and product samples. Three samples from the processing area tested positive for Listeria monocytogenes. Whole Genome Sequencing confirmed that the strain matches the one involved in this outbreak.
Source: FDA
February 21, 2025
The FDA and CDC are investigating a multistate outbreak of Listeria monocytogenes linked to Lyons ReadyCare and Sysco Imperial Frozen Supplemental Shakes. The outbreak has affected 38 people across 21 states, with 37 hospitalizations and 11 deaths reported. Many of the affected individuals were residents of long-term care facilities.
The outbreak was first reported to the FDA on November 25, 2024, after several cases of Listeria monocytogenes were identified in the United States. The FDA's traceback investigation revealed that the affected long-term care facilities had received shipments of the implicated frozen supplemental shakes. Environmental samples collected during the investigation confirmed the presence of the outbreak strain of Listeria.
The outbreak includes cases dating back to 2018, with 20 cases reported in 2024 and 2025. Previous investigations failed to identify the source of the outbreak. The FDA has announced a voluntary recall of certain Lyons ReadyCare and Sysco Imperial Frozen Supplemental Shakes. The investigation is ongoing, and further updates will be provided as new information becomes available.
Source: www.fda.gov/food/outbreaks-foodborne-illness/outbreak-investigation-listeria-monocytogenes-frozen-supplemental-shakes-february-2025
Comments
Comment